For many purchasing managers and business development teams, the quest for a reliable API often feels like sending signals into deep space. You spend hours identifying potential partners, only to be met with weeks of silence or incomplete data. This lack of responsiveness from manufacturers often leads to stalled projects and mounting internal pressure.
At Pharmaoffer, we’ve built a bridge over that gap. Our Smart Sourcing service is more than a form, it is a managed process led by our pharma experts who safeguard your project’s technical requirements while ensuring your timeline stays strictly on track. This blog explains exactly what happens once you click “Submit your request”.

Step 1. Expert inquiry analysis
The journey begins with the details you provide in our structured inquiry form:
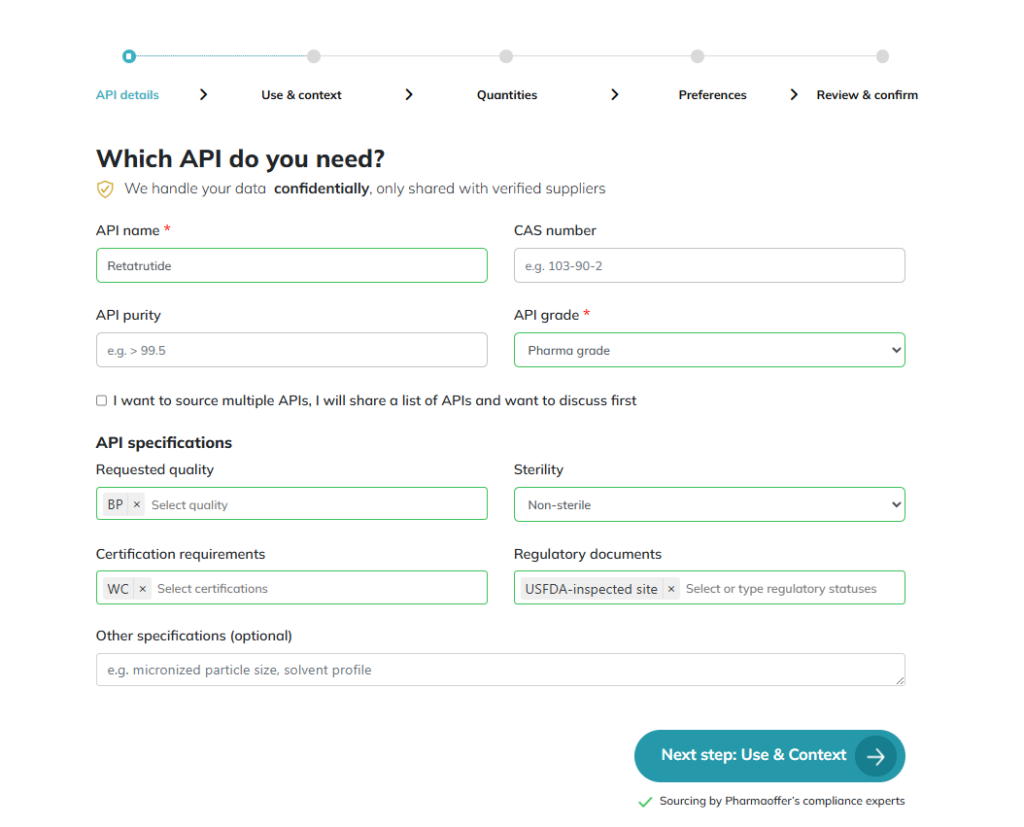
API Details: This includes specific grades and CAS numbers. Our inquiry process begins here: we verify both the CAS and grade to ensure 100% requirement matching from the start.
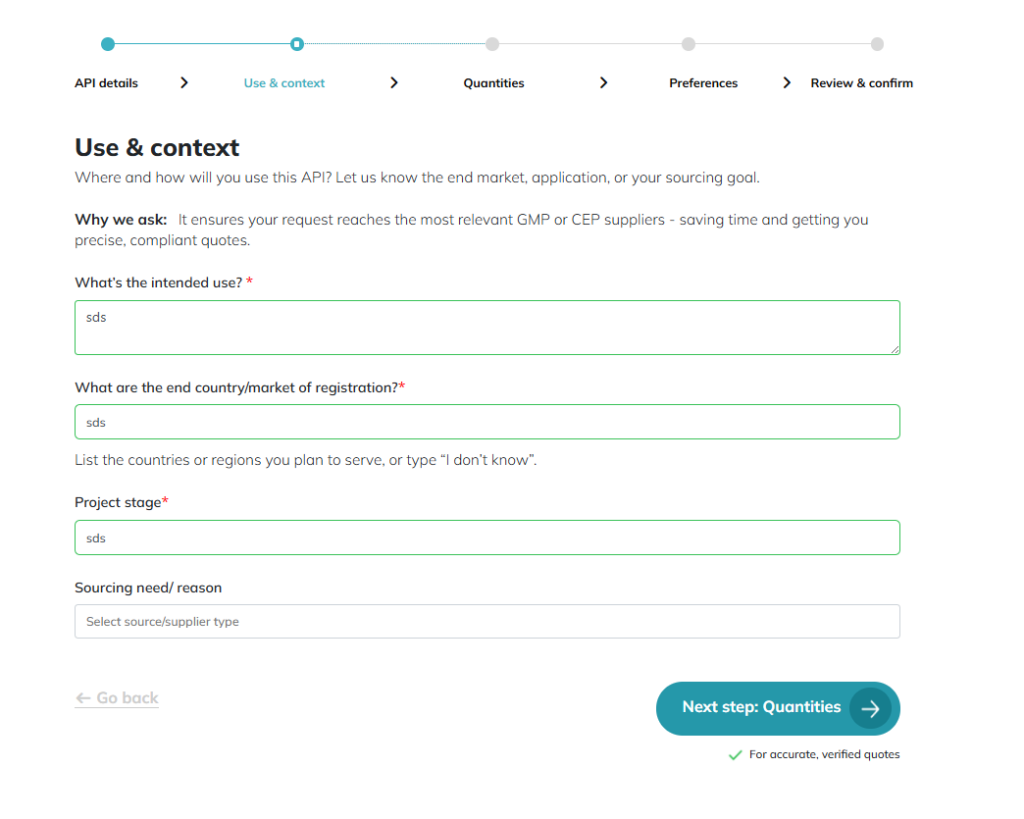
Use & Context: Defining whether the material is for R&D or commercial. The 2nd screen is where you should specify the commercial stage so our team can match you with manufacturers that have the exact regulatory filings required for your market manufacturing.
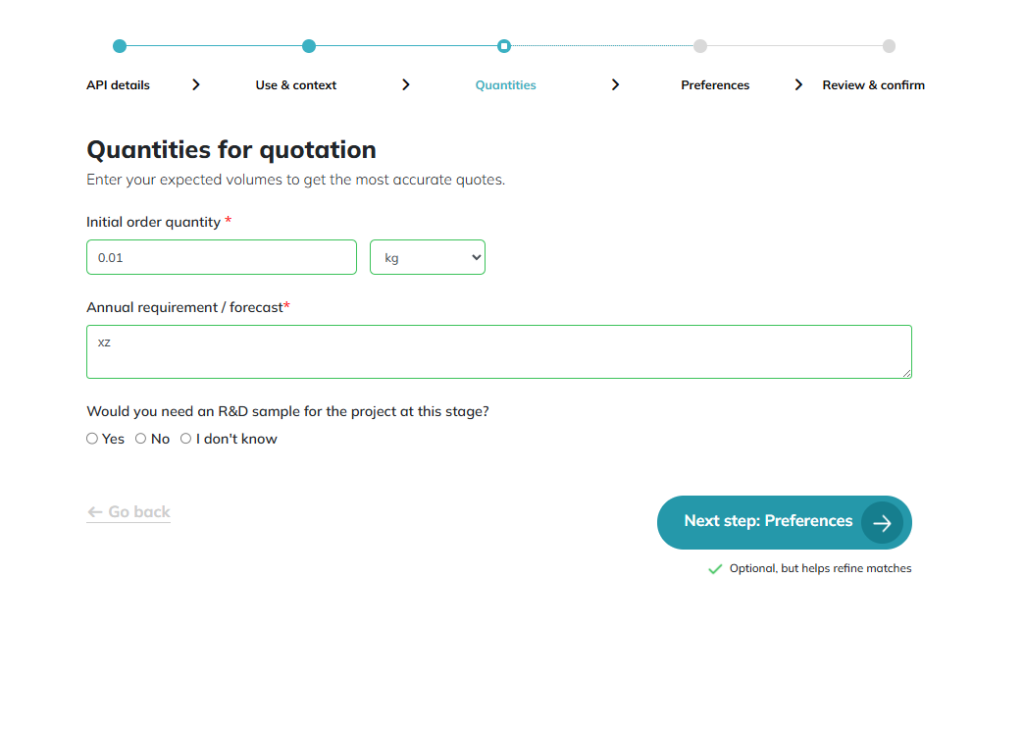
Quantities: Current needs and future projections. Inquiry 3rd screen: Define your scale and scope. Providing precise volumes and regional preferences enables us to target manufacturers that meet your exact logistical and compliance needs.
Preferences: Specific certifications or geographic requirements. Inquiry 4th screen: Final alignment before expert review. Once confirmed, our team optimizes your RFQ to ensure 100% requirement matching and a faster, more professional supplier response.
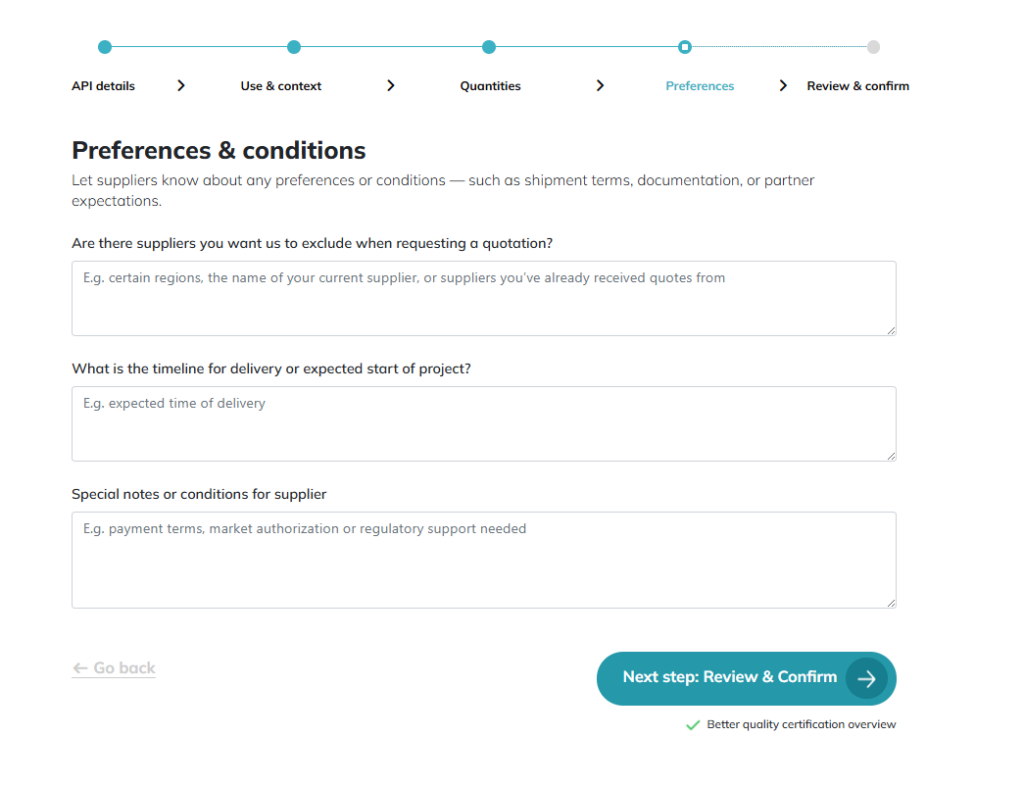
Within one business day, our team reviews these specifics. We refine the inquiry to speak “supplier language,” which significantly improves the quality and speed of the initial feedback we receive.
In this process, you may receive a call or email from us to confirm your requirements or clarify technical details. We observe that being highly specific and detail-oriented at this stage is the single most effective way to secure faster, more targeted matching. By understanding the precise nuances of your project, we ensure a 100% pairing of your requirements with the right manufacturers.
Step 2. Strategic matching and persistence
In the pharmaceutical industry, supply chain relationships remain of the utmost importance for securing long-term reliability. At Pharmaoffer, we prioritize connections with trustworthy manufacturers where we have established a relationship. While anyone can send an email, we leverage these existing partnerships to ensure your inquiry is prioritized by the right stakeholders.
To maintain momentum, we utilize a systematic follow-up process within our network. We take the burden of manual outreach off your desk by managing the communication flow until a clear response is secured. This combination of professional trust and disciplined management helps mitigate the typical industry delays, keeping your sourcing timeline on track.

Step 3. The quality filter (verification)
Before you ever see a name, we perform the “heavy lifting” of document verification. Our team initiates contact within 48 hours to confirm that the suppliers not only have the material but also the valid certifications (GMP, CEP, or specific site registrations) that meet your RFQ.
The Expert Review: Before outreach begins, our Operations Team manually assesses the supplier pool size and potential matches. If fewer than three high-probability suppliers are identified, we notify you to refine the search parameters together. This human-led check ensures we only move forward when there is a high likelihood of success.
If a supplier cannot provide the necessary documentation or fails to respond within our 10-15 day quality window, they are filtered out to protect your timeline. This stage ensures you only spend time evaluating viable, compliant partners.
Step 4. Delivering verified solutions
Once we have validated the supplier’s capacity and documentation, we move to the quote stage. You will receive quotes from manufacturers who have confirmed their readiness to move forward with your specific requirements.
At this stage, we facilitate a direct introduction between you and the manufacturer. Rather than simply passing along data, we ensure a transparent hand-off that allows both parties to align on technical specifics and final terms from the very first interaction. This focused delivery ensures that by the time you receive a quote, the foundation for a reliable partnership is already in place.
Step 5. Success tracking and support
Our commitment to your project doesn’t end with a delivered quote. We track the engagement to ensure the connection is working for both parties.
Because of our managed approach and expert-led verification, we typically observe a high success matching rate for inquiries that move into the quoting stage. Whether the deal is won or you require further market insights, we stay involved to gather feedback and refine your future sourcing strategy.
Transparency & Risk Mitigation: We are committed to quality over quantity. If our rigorous verification process determines that zero suppliers meet your documentation or availability requirements, we initiate a refund immediately. This ensures that your sourcing budget is only spent on viable, verified connections.
Stop Chasing. Start Sourcing.
Waiting on unresponsive suppliers is more than a frustration, it is a risk to your project’s viability. By combining professional rapport with systematic follow-up, Pharamoffer ensures your API requirements are met with the transparency and speed your pipeline demands.
Secure your supply chain with expert-led matching.
Frequently Asked Questions (FAQ)
We prioritize relationships with trustworthy manufacturers where we have established rapport. Before any quote is delivered, our team initiates a verification process to confirm the supplier has valid documentation (such as GMP or CEP) and the necessary manufacturing capacity to meet your RFQ.
We value transparency and project viability. If our initial assessment identifies zero potential matches, or if verification confirms that no suppliers meet your documentation requirements, we initiate a refund. This ensures your procurement budget is only spent on verified, compliant results.
Manufacturers are often overwhelmed with inquiries, which leads to delays. We use a disciplined, multi-channel follow-up system to keep your request at the top of their list. By acting as your professional sourcing arm and leveraging our existing network, we reduce the “response gap” and keep your project timeline on track.
Yes. Once we have validated the supplier’s capacity and documentation, we facilitate a direct introduction. This transparency ensures that both you and the manufacturer are aligned on technical specifics and final terms from the very first conversation.
To achieve a 100% pairing of your requirements, it is vital to be specific. We recommend providing the exact CAS number, the intended use (R&D vs. Commercial), specific quantity projections, and any regional certification preferences.