
TL;DR
- GLP-1 demand growth is pulling strain into capacity, lead times, and supplier reliability – and that increases the value of strong GLP-1 API vendor qualification. FAIR Health
- Semaglutide and tirzepatide remain the baseline today, while the “next wave” (oral GLP-1s, dual or triple agonists, longer-acting options) will change how buyers should plan sourcing and risk controls. (Wharton et al., 2023)
- GLP-1 API sourcing is increasingly shaped by pipeline change, not just today’s demand – especially as oral and multi-agonist programs progress (Wharton et al., 2023), (Jastreboff et al., 2023).
- Procurement teams that win in this category tend to move earlier on supplier selection, qualification readiness, and continuity planning – before the “rush” hits.
- Counterfeit and unapproved versions are a real sourcing risk signal – it’s another reason procurement and QA need tighter verification and traceability requirements. (FDA; WHO)
- If you want fewer delays later, treat GLP-1 API supply chain planning like a program (not a one-off purchase) and build a dual-sourcing strategy with clear quality gates.
If you’re responsible for buying APIs or running sourcing projects, GLP-1 is probably already on your radar, not only because demand is big, but because the supplier and risk landscape is changing fast.
The most useful way to think about GLP-1 API sourcing right now is simple: reduce cycle time without compromising qualification. The winners won’t be the teams who move fastest at any cost. Winners will be the teams who build supply continuity early, qualify smarter, and negotiate using data rather than assumptions.
At this very moment, many forces, or therapy drugs in this case, are colliding. Demand is rising quickly, and the development landscape is shifting toward differentiated assets (oral delivery, dual and triple agonists, less frequent dosing). That combination can tighten supply, raise quality expectations, and increase the cost of being single-sourced.
This blog focuses on what purchasers and cross-functional sourcing teams can do now to reduce surprises later.
Why GLP-1 demand is now a sourcing priority
Obesity and overweight are already widespread, and prevalence is still trending upward. The WHO reports that in 2022, 43% of adults were overweight and 16% were living with obesity.
Forward projections reinforce that this is not a short-cycle wave. The World Obesity Federation’s 2025 Atlas projects that by 2030, roughly half of adults (age 20+) could have “high BMI” (overweight or obesity, combined).
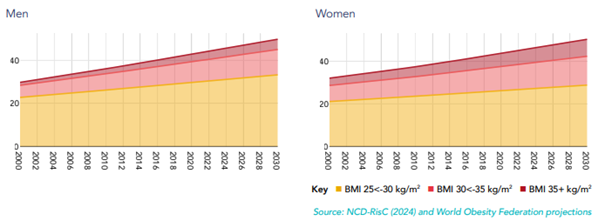
On the utilization side, FAIR Health reported a sharp increase in GLP-1 prescribing between 2019 and 2024, highlighting how quickly demand dynamics are changing.
Financially, major market forecasts are now large enough that they influence investment, manufacturing allocation, and competitive behavior across the supply chain. Morgan Stanley Research estimated the global obesity drug market could reach $150B at its peak in 2035.
For purchasers, the takeaway is simple: this is a category where sourcing lead time, qualification readiness, and continuity planning will increasingly differentiate who can supply reliably.
Semaglutide API: the demand anchor you have to plan around
Semaglutide is still the key reference point in modern GLP-1 therapy. It has established efficacy in obesity and is widely used, which makes it a major demand anchor for the API market and a baseline comparator for newer candidates. (Wharton et al., 2023)
In the STEP 1 trial (68 weeks), once-weekly semaglutide 2.4 mg plus lifestyle intervention produced a large mean weight reduction versus placebo, helping define expectations for this class. (Wharton et al., 2023)
What it means for GLP-1 API supplier selection
First, semaglutide tends to pull capacity and attention across the supply base. Even if you’re not sourcing semaglutide itself, its market gravity can affect lead times and availability for adjacent peptides, intermediates, and specialized manufacturing slots.
Second, semaglutide has become a counterfeit target. Both the WHO and FDA have issued alerts about falsified or counterfeit Ozempic (semaglutide) being detected, including instances that entered regulated supply chains. That’s a serious reminder that vendor qualification and traceability are not “nice to have” in GLP-1 API procurement, they are part of risk management. (WHO; FDA)
For your workflow, this pushes you toward tighter controls: more rigorous supplier verification, stronger chain-of-custody expectations, and clearer red lines on documentation quality.
Tirzepatide API: changing procurement expectations
Tirzepatide matters for GLP-1 API sourcing strategy because it pushed expectations upward for weight loss outcomes and intensified competition across the metabolic space. In the SURMOUNT-1 trial, once-weekly tirzepatide produced substantial weight reduction versus placebo over 72 weeks. (Aronne, L.J. et al. 2024)
Regulatory momentum also reinforced the scale of demand. The FDA approved Zepbound (tirzepatide) for chronic weight management in adults with obesity or overweight (with at least one weight-related condition). (FDA)
From a procurement perspective, tirzepatide does two things:
- It increases urgency among sponsors and manufacturers to secure reliable supply earlier, because commercial demand can build quickly once a product is established.
- It also raises the baseline for “what’s good enough,” which accelerates next-generation programs and tightens the overall market for qualified capacity. The more the market expects stronger efficacy, the more pipelines fill with newer assets – and the more competition you see for high-quality manufacturing slots.
If you’re building a GLP-1 API vendor qualification approach, tirzepatide is a strong example of why you should treat supplier continuity planning as an early-phase activity, not something you start after demand spikes.
What “next wave GLP-1” really means
When people say “next wave,” they’re usually pointing to three shifts:
One shift is oral delivery. Orforglipron is described as an oral, non-peptide, small-molecule GLP-1 receptor agonist in late-stage development, positioned partly on convenience and simpler administration compared with oral semaglutide’s dosing constraints. (Wharton et al., 2023)
Second shift is dual and triple agonists, designed to move beyond “single mechanism” GLP-1 effects. Retatrutide is positioned as a triple agonist (GLP-1, GIP, glucagon receptors), and the report frames it as a potential competitor to established injectable therapies depending on phase 3 outcomes. (Jastreboff et al., 2023). Find the latest data intelligence for orforglipronand retatrutide API.
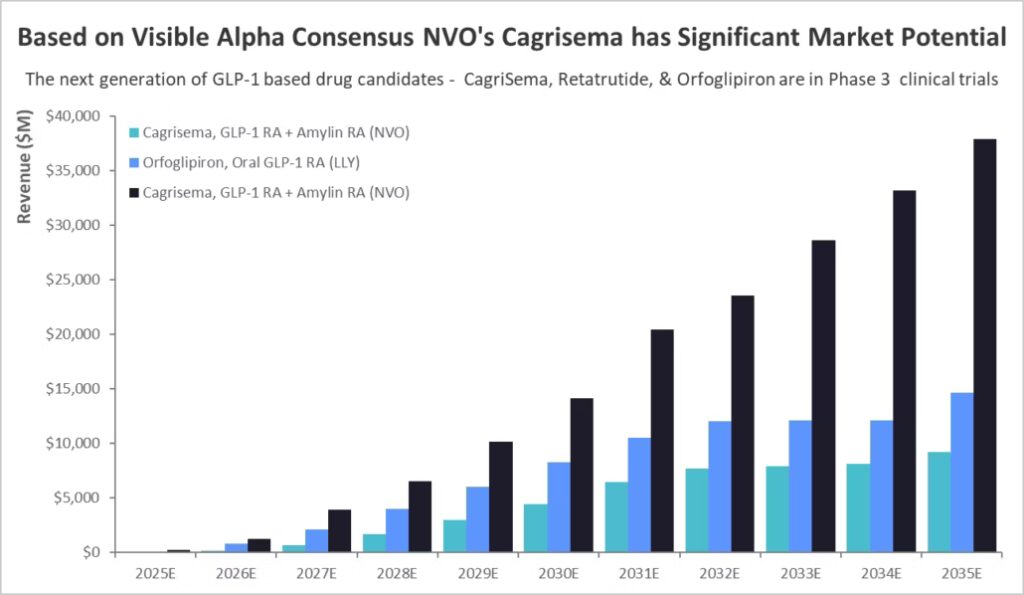
Third shift is the focus on tolerability, adherence, and real-world durability. This means the commercial winners may not be determined only by peak efficacy, but also by discontinuation rates, dosing convenience, and payer dynamics. In the Drugs to Watch Report, Clarivate, frames this broader trend as a race to reinvent weight loss, reflecting how crowded and fast-moving the obesity pipeline has become.
Importantly, not every program makes it. Pfizer’s discontinuation of danuglipron after a trial safety signal is a reminder that pipelines shift – and when they do, demand and supplier priorities can shift quickly too.
The takeaway is simple: For procurement, these shifts influence what “good supplier strategy” looks like. You’re not only picking an API source, you’re choosing how resilient your supply chain is when formats and demand patterns change. That means your GLP-1 API sourcing strategy should be built to absorb change.
What changes for GLP-1 API sourcing
1) Continuity becomes the KPI
With GLP-1, a single-source mindset is a risk multiplier.
Even if you have a preferred vendor, you want continuity planning that assumes disruption can come from capacity constraints, commercial allocation decisions, or unexpected safety and program changes.
A practical way to think about it is: you’re not qualifying a supplier – you’re qualifying your supply chain. That usually means putting dual sourcing strategy for APIs on the table earlier than you might for slower categories, and aligning internally on what “acceptable equivalence” looks like before you’re under time pressure.
2) Qualification speed becomes a competitive advantage
GLP-1 API vendor qualification often slows down for one reason: documentation isn’t ready, or it isn’t consistent.
If you want to compress cycle time without compromising qualification, your best lever is to standardize what you request upfront and insist on completeness before you invest heavy time.
In practice, you’ll want clean, comparable packs early: GMP status evidence, CoA expectations, traceability information, and anything else your QA and regulatory teams require for your specific market and dosage form.
When you do this early, procurement stops chasing documents later. You get faster internal alignment and fewer late-stage surprises.
3) Risk controls need to be explicit, not assumed
High-demand categories attract noise: brokers who can’t prove origin, “manufacturer” claims that don’t stand up to verification, and offers with pricing that looks great until you ask basic questions.
You don’t need to assume bad intent, but when counterfeit incidents rise, it’s a market signal that demand pressure is creating vulnerabilities. Regulators have issued warnings and alerts on falsified semaglutide products, and FDA communications highlight how counterfeit products can contain the wrong ingredients, incorrect strength, or harmful components. (FDA; WHO).
A simple mindset shift helps: treat supplier verification as part of purchasing, not a separate QA clean-up step.
Quality and documentation: Where GLP-1 sourcing teams get slowed
GLP-1 sourcing delays are often created by “documentation reality,” not outreach volume.
Purchasers and QA teams typically lose time on:
- Incomplete audit packages or unclear GMP evidence.
- Gaps in traceability – for example around manufacturing site clarity, subcontractors, or material flow.
- Misalignment on what “acceptable” means for change control, deviations, and ongoing communication.
You do not need to turn procurement into QA, but you do need to agree on minimum gates early and keep them consistent across suppliers so comparisons are meaningful.
A practical mindset: aim for fewer suppliers in your shortlist, but make each one “qualification-ready,” not just “commercially responsive.”
What this changes for supplier strategy: What to do for GLP-1
If you’re building a supplier portfolio, this is the moment to tighten your selection criteria.
GLP-1 API supplier selection should prioritize three things:
Can they reliably supply at the volumes you may need.
Can they support your qualification timeline with complete documentation.
Can they demonstrate traceability and a credible manufacturing footprint.
If any one of those is weak, you can still engage – but you should engage with the right expectations and safeguards.
Where procurement teams get stuck (and how to avoid it)
Most GLP-1 API sourcing delays come from a predictable pattern.
You start outreach wide.
You receive inconsistent responses.
You spend weeks normalizing data, chasing documents, and trying to compare offers that were never comparable.
Then the process becomes slow, even when the category is moving fast.
If you want to avoid that, the fix is not “more supplier outreach.” It’s a better workflow.
The workflow that keeps GLP-1 API sourcing moving
- Define your API requirement and timeline clearly enough that suppliers can quote without multiple clarification loops.
- Pre-screen suppliers based on qualification readiness, not just response speed.
- Request quotes in a way that makes offers comparable (incoterms, lead time assumptions, packaging, documentation included, and validity period).
- Shortlist based on total sourcing risk, not only price per kg – then proceed with the qualification steps you already trust.
This is also the point where program management pays off: track assumptions, dates, and supplier commitments in one place so your internal stakeholders (QA, regulatory, manufacturing) aren’t working from fragments.
How Pharmaoffer supports this workflow
Pharmaoffer is built for sourcing teams who want speed and control, not noise.
Smart sourcing service – verified quotes in days
If your goal is to move faster without cutting corners, the best support is the support that reduces uncertainty. The Smart Sourcing Service is designed to reduce cycle time without lowering qualification standards.
- You send your API specifications to our expert team once.
- We validate the request and start targeting outreach within a network of verified suppliers.
- You receive 3-5 qualified supplier offers you can actually compare.
Suppliers reply within a few days, and your data is handled confidentially and shared only with verified partners.

Search in our free verified supplier marketplace and request a quote directly to any supplier without limitations.
Sources:
- World Health Organization (WHO) – Obesity and overweight facts and prevalence.
- World Obesity Federation – World Obesity Atlas 2025 projections (through 2030).
- FAIR Health – GLP-1 utilization trends (2019-2024).
- Morgan Stanley Research – Obesity drug market forecast to 2035.
- NEJM – Orforglipron trial publication (Wharton et al., 2023).
- NEJM – Retatrutide trial publication (Jastreboff et al., 2023).
JAMA – Tirzepatide trial publication (Aronne, L.J. et al., 2024). - Pfizer press release – Discontinuation of danuglipron (April 14, 2025).
- Clarivate – Drugs to Watch 2026 overview and obesity trend framing.
FAQ’S
What is the difference between GLP-1 API procurement and GLP-1 finished dose sourcing?
API procurement focuses on qualifying and securing the active ingredient supply chain (quality, traceability, continuity). Finished dose sourcing adds formulation, fill-finish, packaging, and additional regulatory and tech-transfer complexity.
How do I find reliable GLP-1 API suppliers?
Start with qualification readiness: GMP evidence, clear site ownership, consistent documentation, and responsiveness to technical questions. Then compare suppliers using the same checklist so “fast replies” do not win by default.
What should a GLP-1 API supplier audit checklist include?
At minimum: GMP status, site scope, change control approach, deviation handling, traceability, analytical capabilities, and clarity on subcontracting.
How should I think about counterfeits and verification in GLP-1 API sourcing?
Treat verification as a process, not a document. Confirm supplier identity, site reality, traceability, and consistency across documents and communications – and avoid making decisions based on price alone.
How do I know if my GLP-1 is real?
Only use GLP-1 obtained through a licensed pharmacy and prescriber. Avoid online “vials” or “research” products – that’s where counterfeits and mislabeling risk spikes.
How do I check if semaglutide or tirzepatide is authentic?
Confirm the supply route first (licensed pharmacy). If anything looks off (packaging, labeling, unexpected side effects), stop and ask a pharmacist or clinician to verify.
How do I know if my GLP-1 API is authentic?
Treat “real” as proven identity, purity, and traceability. Use qualified suppliers plus independent testing and documented batch traceability – not just a CoA screenshot.
Why are GLP-1 compounds still being sold online?
Some are compounded under limited rules, but many are unapproved or illegally marketed. If it’s sold direct-to-consumer as “research” or without a prescription, it’s a red flag.
Why are GLP-1 compounds still being sold online?
Some are compounded under limited rules, but many are unapproved or illegally marketed. If it’s sold direct-to-consumer as “research” or without a prescription, it’s a red flag.
Are GLP-1 drugs being banned?
Usually not the drug class. Regulators mainly restrict counterfeit, unapproved, or illegal distribution routes, and sometimes tighten compounding rules when supply stabilizes.
What are the risks of GLP-1 medications?
Common issues are GI side effects; rarer risks exist and should be discussed with a clinician based on your history.
What is the controversy with GLP-1?
It’s mostly about access and pricing, safety discussions at scale, and the growth of counterfeit or unapproved products.
What would cause a GLP-1 drug to fail?
Not meeting efficacy endpoints, safety or tolerability signals, or failing to differentiate in a crowded market.
What happens if you stop GLP-1?
Weight regain is common for obesity indications, and for diabetes indications stopping can worsen glycemic control. Don’t stop without medical guidance.





