What are APIs?
(Active Pharmaceutical Ingredients)

Ammar Badwy | Posted on September 21, 2022
Introduction
The first thing that comes to your mind when discussing the pharmaceutical industry is companies that produce medicines. All over the globe, there are lots of companies that make medicines.
From big pharma companies such as Novartis, Pfizer, and Johnson & Johnson to local pharmacies, which are, in some cases, able to make medicines on a patient level. And, of course, everything in between. All these medicine makers need APIs to produce medicines. But what are APIs?
We made a video about it, which you can find below.
(text continues under the video)

What are APIs?
People take medicines to cure, diagnose, treat, or prevent a disease. The most medication contains a couple of ingredients. The most important is the API, which stands for Active Pharmaceutical Ingredient. This is the actual substance in the medicine that has to do the work in your body.
For example, the API in Aspirin is “acetylsalicylic acid.” This API will try to fight the pain of your fever or headache. Some medicines carry the name of the API. For instance, the API of Paracetamol is “paracetamol“. To make a pill, you’ll need more ingredients than just the API. That’s where excipients come in. These ingredients are used to give the pill, for example, volume, a sweet flavor, or a color.
The term API is not only used in the pharmaceutical industry but also in the web developers community the term API is common. For them, however, it stands for “Application Programming Interface.”
How and where are APIs made?
The API market alone is already a multi-billion dollar market. APIs are mainly produced in large production plants. It’s a complex chemical process of several steps. It’s made from starting materials which can be a raw material or intermediate. Back in the days, pharma companies made and formulated the API and medicine in their home countries.
Over the past few decades, many companies have outsourced their pharmaceutical ingredients manufacturing (API) process to Asia to cut costs. Today, India and China are the largest API-producing countries. Some of the largest suppliers are Aurobindo, Dr. Reddy’s, Cipla and SUN Pharma. They are all founded or based in India. But, since most starting materials are made in China, China is the root source of most APIs in the world. APIs are often made in powder form and sold in large quantities to drug manufacturers.
Quality of APIs (active pharmaceutical ingredients)
Medication is, of course, intended to help people. So, we need to know the ingredients, such as the API, are safe to use. No matter where an API is produced, it has to meet the safety and quality criteria of the country where end users are located. That means drugs sold in the EU need to meet the strict safety and quality standards of the European Medicines Agency, and those sold in the US need to meet the regulations issued by the U.S. Food and Drug Administration (FDA).
Companies in the entire API supply chain get inspected by their local government. Foreign governments instances could also inspect them, and third-party companies do audits of each other companies.
When everything is up to code, companies are given a certain certificate, such as GMP or a written confirmation, so other professionals in the industry know that that specific company complies with the industry standards without them having to inspect or audit themselves. When the inspection fails, the companies will be issued a warning, and pharmaceutical buyers won’t be able to buy there until the issues are resolved and the company is re-inspected.
Not only the factory or production facility is inspected, but laboratories also analyze every batch of produced API; depending on the situation, it is even possible that one specific batch is analyzed multiple times: For example, by the company that produced it, by a 3rd party laboratory, by the trader who buys it, and by the hospital that will use it.
The difference between QA and QC
Also, every pharmaceutical company has an entire team dedicated to quality assurance and quality control. This team can consist of pharmacists, chemists, biologists and other trained and qualified personnel. You could say that the first team, quality assurance, ensures everything is fine before the production process starts, while quality control ensures everything is performed according to plan.
Nonproprietary names: USAN/INN
While the number of available prescription or over-the-counter drugs (OTC) under different names increases daily, the situation differs in the case of APIs. Considering how expensive the steps to introducing a new drug to the market are, there isn’t a great number of new APIs being introduced. An API is sold under only one name: the nonproprietary name.
To avoid confusion in this market, which potentially would have dangerous consequences, WHO, in cooperation with national drug safety agencies worldwide, came up with a list of non-proprietary names back in 1953. The organization revises the list regularly, and you can send them a request for new International Nonproprietary Names (INN).
So, everywhere in the world, Ramipril is sold as Ramipril, and that API can’t be bought under any other name. Drug manufacturers can sell their drugs under any name they want, but they must put the API name right under the brand name.
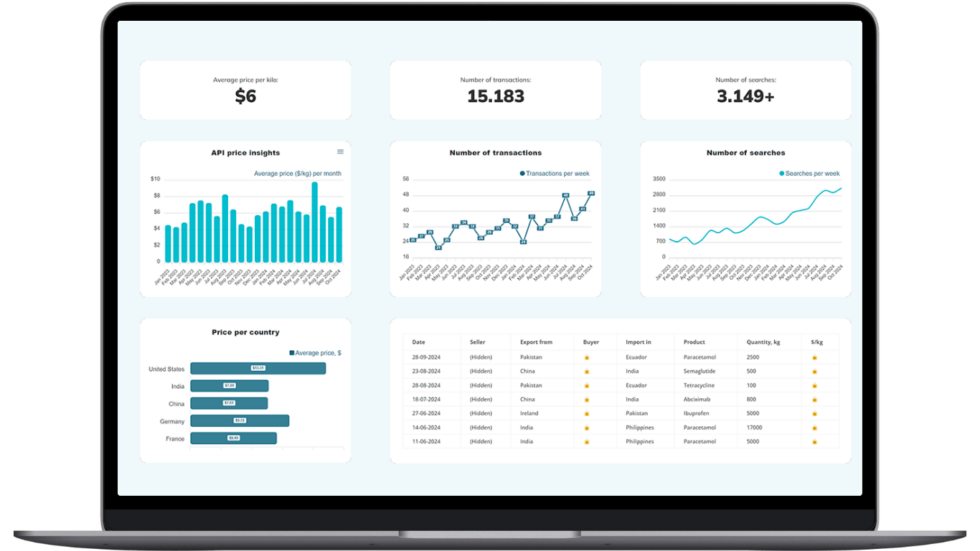
Make Smarter API Decisions with Data
Access exclusive insights on global API pricing, export/import transactions, competitor activities and market intelligence.
FAQ
What is an API in Pharmaceuticals?
API stands for Active Pharmaceutical Ingredient. It is the key substance in a medication that performs the main action of treating, diagnosing, or preventing a disease. For example, in Aspirin, the API is "acetylsalicylic acid."
How Do APIs Work?
APIs are designed to interact with specific biological pathways to produce a therapeutic effect. For instance, the API in Aspirin, acetylsalicylic acid, works by inhibiting enzymes that produce pain-signaling molecules.
Where Are APIs Manufactured?
APIs are primarily manufactured in large production facilities. Countries like India and China are leading in API manufacturing due to cost-efficiency.
How is API Quality Ensured?
APIs must meet stringent safety and quality criteria set by health authorities like the European Medicines Agency and the U.S. Food and Drug Administration (FDA). Facilities are regularly inspected, and batches of APIs are analyzed multiple times for quality assurance.
What is a Nonproprietary Name in API?
A nonproprietary name is the standard name under which an API is sold globally. It is regulated by the World Health Organization to prevent confusion and enhance safety.






Check out all other blogs here!